The story of Remdesivir
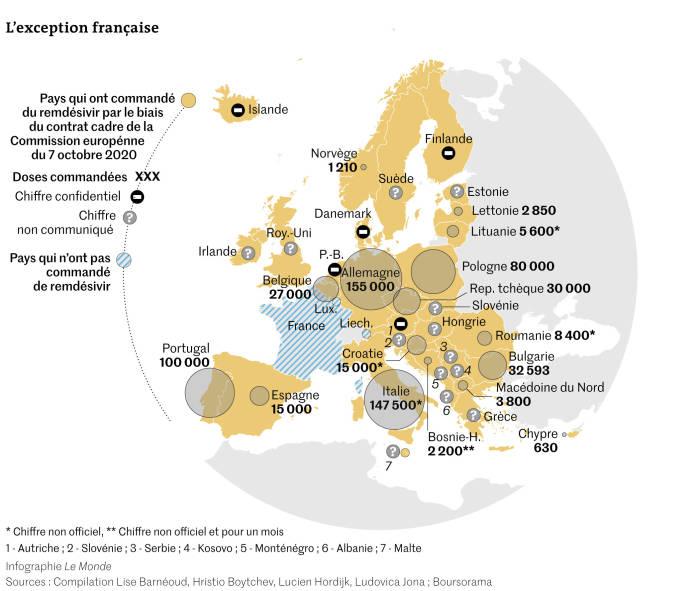
On October 7th 2020, the European Commission signed a contract with the company without negotiating on the price, set by Gilead at €2070 per treatment (6 doses). Only a week later, the WHO published the largest study carried out so far on the drug, which shows its ineffectiveness, both in reducing the death of patients and their hospitalisation.
This investigation has been conducted by a cross-border team of journalists from Italy, France, Switzerland, Germany and the Netherlands. According to their sources, a great part of member states' authorities were pressured to buy immediately after the EU agreement, due to the scarcity of Remdesivir.
The team asked the authorities of each of the 37 member countries of the agreement (all EU Member States and 10 associated states such as Bosnia-Herzegovina and Albania) for the number of doses already purchased, obtaining data from 17 states which bought for at least 226 million euros (plus the 70 million previously spent by the EU for around 30,000 doses). Major buyers are not the richest countries in the EU, on the contrary. France did not buy a single phial, while Portugal and Bulgaria bought more than Germany in proportion to their inhabitants.
Photo image: Le Monde
Consultant: Nicoletta Dentico